|
The
APTA Vision Statement for Physical Therapy 2020 contains an
element which suggests the application and integration of evidence
to guide clinical decision making to provide the best practice for
the patient/client. In order to obtain this evidence it will be
essential to continue to conduct and evolve research but in an
ethical manner. In this article I will
review the ethical issues in human experimentation.
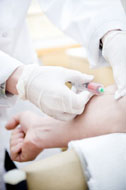
Scientific research requires studies on
human subjects or on fluids drawn from human subjects. New drugs or
technologies are normally tested on animals first & as the research
progresses, products are tested on humans before they could be used
by physicians in clinical practice. Since the tests/drugs could
cause harm to humans, experimentation on humans poses to be an
ethical concern. Another ethical concern in research/experimentation
is what it does to the people or subjects of testing. This includes
embryos & fetuses which are part of studies & what happens of them
is an ethical concern since embryos & fetuses are human life &
harming it without sufficient reason is not moral. According to
Ethical & Religious Directives (1), “No one should
be the subject of medical or genetic experimentation even if it is
therapeutic, unless the person or surrogate first has given free and
informed consent. In instances of non-therapeutic experimentation,
the surrogate can give this consent only if the experiment entails
no significant risk to the person’s well-being. Moreover, the
greater the person’s incompetence and vulnerability, the greater the
reasons must be to perform any medical experimentation, especially
non-therapeutic.”
History shows that subjects used for human experimentation were
mostly disenfranchised like orphans, mentally retarded, prisoners &
charity patients (5). Even the most defenseless are not safe from
those researchers who possess no heart. The mentally retarded are
easy targets for human experimentation. However it sometimes gets
difficult to get human subjects for experimentation especially for
those involving high risk or pain. In 1930s researchers found a
solution to the shortage of human subjects for experimentation - don’t ask
for consent (6).
The recent moral awareness about medical research & laws regulating
them has developed as a reaction to ill treatments of human beings
in research in the past. History of medical research or
experimentation throws light on how it became unethical when desire
to achieve medical & scientific progress overshadowed moral
sensitivity to the well being of patients involved (1). A look at well
known abuses in research is important in understanding the current
concerns in ethics in medical research. The Tuskegee Syphilis Study,
Cruel Nazi experimentations in German Camps, and the Willowbrook
School Hepatitis study are just some examples of atrocities done on
humans in experimentation with no regards to morality or ethical
consideration.
As a result of the international outrage over Nazi Medical
experimentation, judges at Nuremburg set forth Principles for Medical
Research, the first 10 of its principles are known as the Nuremburg Code
(4). This Code
was published by the U.S. Government Printing Office in 1949. United
States
medical researches were to abide by the Code from then onwards but
unfortunately sometimes they did not (7).
There was a growing sense of discomfort about this & many other
studies which was instrumental in developing standards of research &
human experimentation. Henry Beecher, a professor of anesthesia at
Harvard Medical School published an article in The New England
Journal of Medicine identifying 22 research projects where
researchers failed to provide adequate information to their subjects
or were successful in obtaining voluntary consent. These studies
were funded by various governmental agencies & pharmaceutical
companies. Reports of the studies were cited in journals like
Journal of American Medical Association, The New England Journal of
Medicine, and Journal of Clinical Investigation & Circulation.
Beecher also identified many other studies with questionable ethical
values. His work made an immediate impact .The government set forth
rules & regulations raising the moral values of human
experimentation. Later Pappworth & Jay Kartz presented many papers
revealing the unethical conduct of experimenter/researchers.
Early Federal Initiatives to regulate guidelines on human
experimentation are tracked to the time even before Beecher. In
1966, the Surgeon General issued the STATEMENT OF POLICY on clinical
investigations using human subjects requiring approval of research
funded by the public Health Services. After Beecher's article was
published, the Dept. of Health & Education & Welfare issued the
Institutional Guide to DHEW Policy on Protection of Human Subjects
(1971) (7)- this emphasized informed consent & institutional review.
By now, these committees reviewing research proposals were called
Institutional Review Boards.-IRB.
The U.S. Congress passed the National Commission for protection of
human subjects of Biomedical & Behavioral Research which was
instrumental in developing medical ethics during that decade. The
National Commission also adopted the principles of ethics advocated
by William Frankena- first being Justice -understood as equality &
obligation to treat everyone equally & fairly. The second being
beneficence - understood as general good-doing greatest good for
greatest number. Autonomy i.e. respect to ability of patient to
decide for himself was the other principle. This model of ethics is
used to analyze if a moral agent behaves morally well. The second
report of the National Commission was the IRB. The IRB's main
function is to protect the needs of human subjects in human
experimentation.
Along with these directives there were other documents prepared to
govern the rights of human experimentation including The Helsinki
Declaration and the Belmont Report.
Science has made many fascinating advances. This has all been
possible with numerous trials & experimentations. Human
experimentation is an essential step to test effectiveness of a
chemical or technique to help develop effective treatments for
individuals. I believe that experimentation can be done on willing
human subjects but with strict rules to ensure just treatment &
respect to all human life. Following are a few regulations that may
serve as a guideline to guarantee the above:
- There is a possible harm to subjects of human experimentation &
we should keep in mind that everyone should be humane in dealing
with them.
- Experimenters & physicians should not be tempted to feed their
curiosity at the cost of harming someone else. They should follow
the code of human experimentation appropriately. They should inform
an individual completely about the trial with its consequences. They
should take full responsibility to treat them in case of infection
or disease.
- Subjects should be allowed to retract at any time they want & be
honored the usual treatment option if they want to leave the study
at any time.
- An Institution Review Board (in accordance to FDA regulation)
should oversee the proceedings of human experimentation to ensure
subjects are treated ethically & morally & the experiment is
conducted in an ethical manner. An IRB is a board, committee, or
other group formally designated by an institution to review, to
approve the initiation of, and to conduct continuing review of
biomedical research involving human subjects in accordance with FDA
regulation.
Ethical issues arise from emergence of new technologies that can be
harnessed. It’s true that no amount of experimentation on animals in
labs can be completely reliable in predicting outcome of disease or
intervention on humans. Doctors have recognized the importance of
human experimentation since the days of Hippocrates, though the
ancient Greeks used it to benefit individual patients rather than
science itself or any profit-driven industry. There is temptation
for experimentation to go too far often jeopardizing the quality of
human life involved. Curiosity is a powerful human motivation which
can lead well-meaning people to actions that are harmful, and even
fatal, as a look at the most extreme cases clearly shows (2).The
growth of technology only makes vigilance more necessary. As science
is growing it is getting more & more difficult to keep a control
over it. Scientists have to learn to restrain & inhibit their pulse
of curiosity that could cause potential harm to innocent human
subjects. Study of ethics in human experimentation helps to
understand where a draw a line when it comes to dealing with humans.
References
1)
http://www.ascensionhealth.org/ethics/public/issues/human_experiment.asp
2) http://www.pcrm.org/resch/humres/humanexperiments.html
3)
http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1816110
4) http://www.rbs2.com/humres.htm
5)
http://www.associatedcontent.com/article/203447/when_is_human_experimentation
6) http://www.naturalnews.com/019193.html
7) Practical Decision Making in Health Care Ethics: Cases and
Concepts Feb 2000 - Raymond J. Devettere
Last revised: July 10, 2009
by Puja Panjwani, PT
|